AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Guideliner vascular solutions1/4/2024 
New clinically unique solutions for vascular diseases to physicians worldwide. Formed in 1997, VSI is a medical device company focused on bringing BACKGROUND Vascular Solutions, its GuideLiner® Product, and the Patents-in-Suit 8. Venue is proper in the District of Minnesota pursuant toĢ8 U.S.C. Scientific maintains places of business within the State of Minnesota and this district Boston Scientific regularly conducts business in the State of Minnesota and this district and VSI’s cause of action arises directly from Boston Scientific’s infringing actions by manufacturing, marketing and selling the infringing Guidezilla™ product in the State of Minnesota and this district. The Court has personal jurisdiction over Boston Scientific, because Boston This Court has jurisdiction over the subject matter of this action underĢ8 U.S.C. This action arises under the Patent Act, 35 U.S.C. Scimed Place, Maple Grove, Minnesota 55331, and in numerous other states and countries. Boston Scientific also maintains a place of business and manufacturing operations at Two 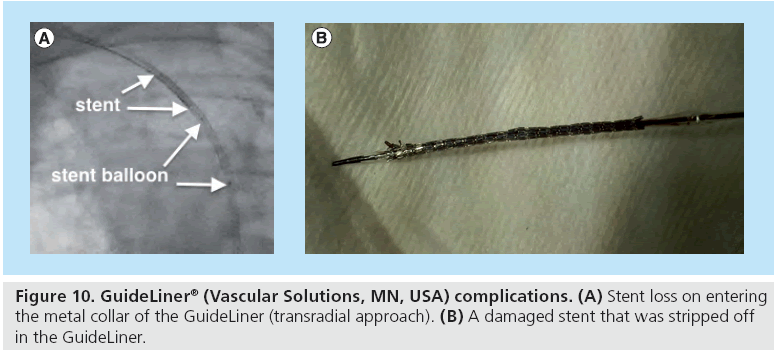 
Headquarters at One Boston Scientific Place, Natick, Massachusetts 01760. Defendant Boston Scientific is a Delaware corporation, with its corporate VSI is the owner by assignment of the patents-in-suit. Plaintiff VSI is a Minnesota corporation, with its principal place of businessĪt 6464 Sycamore Court North, Maple Grove, Minnesota 55369. C), all entitled “Coaxial Guide Catheter for Interventional Cardiology Procedures” (collectively, the “patents-in-suit”). This is a patent infringement action to stop Boston Scientific’s infringement (“VSI”), for its complaint against Boston Scientific Corporation (“Boston Scientific”), states and alleges as follows: 1. Boston Scientific Corporation, Defendant. The catheters may be used to facilitate placement or exchange of guidewires and to subselectively infuse/deliver diagnostic and therapeutic agents.UNITED STATES DISTRICT COURT DISTRICT OF MINNESOTA The Turnpike catheters are intended to be used to access discrete regions of the coronary and/or peripheral vasculature. All three versions feature a hydrophilic coating on the distal 60cm of the catheter for smooth advancement. The new Turnpike Gold version uses the same shaft but features a threaded, gold-plated tip designed for rotational advancement in cases in which a 0.014” guidewire has been advanced through a lesion but even the smallest balloon catheter is unable to be delivered over the wire. The new Turnpike Spiral version uses the same shaft and tip configuration but adds an outer coil attached to the distal 2cm of the shaft, which is useful in procedures in which physicians need additional rotational advancement of the catheter through tortuous anatomy. The original version of the Turnpike, launched in January, extends the catheter’s braid and dual-layer coil into a soft, radiopaque distal tip which, following a non-tapered polymer-only section, gradually tapers to a smooth distal edge. The single-lumen catheters are constructed in five layers: two layers of polymer encapsulating both a braid and a dual-layer coil. 
The design of these catheters allows for excellent tracking in tortuous vessels and superior passage through difficult stenoses.”Īll three versions of the Turnpike are over-the-wire catheters with an advanced shaft design that provides superior tracking and advancement over a 0.014” guidewire. “I have been very impressed with the new Turnpike catheters and have incorporated these devices into my practice. “A major focus of my practice is the treatment of complex cases, and these interventions require specialised devices to navigate in highly tortuous vessels and through extremely tight plaque lesions,” Spratt stated. One of the early adopters of the Turnpike catheters is James Spratt, consultant cardiologist at Forth Valley Acute Hospitals, UK. Since launching our GuideLiner guide extension catheter in 2009, Vascular Solutions has been focused on providing tools for use in the most challenging interventions, and we expect our Turnpike catheters to make a significant added contribution in this rapidly growing clinical specialty worldwide.” “At a time when physicians are performing a greater number of complex interventional procedures, we are pleased to broaden our Turnpike product line with two new devices that provide unique clinical benefits. “Physicians have responded enthusiastically to our launch of the original Turnpike catheter, which is turning out to be one of our most successful new product launches in recent years,” said Howard Root, chief executive officer of Vascular Solutions. The original version of Turnpike was introduced in January 2015, and now Vascular Solutions has added the Turnpike Spiral and Turnpike Gold versions to the product line. Vascular Solutions has launched two new versions of its Turnpike catheters for use in complex coronary and peripheral interventions.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
